Moderna to Begin Testing Its Vaccines in Babies as Young as Six Months
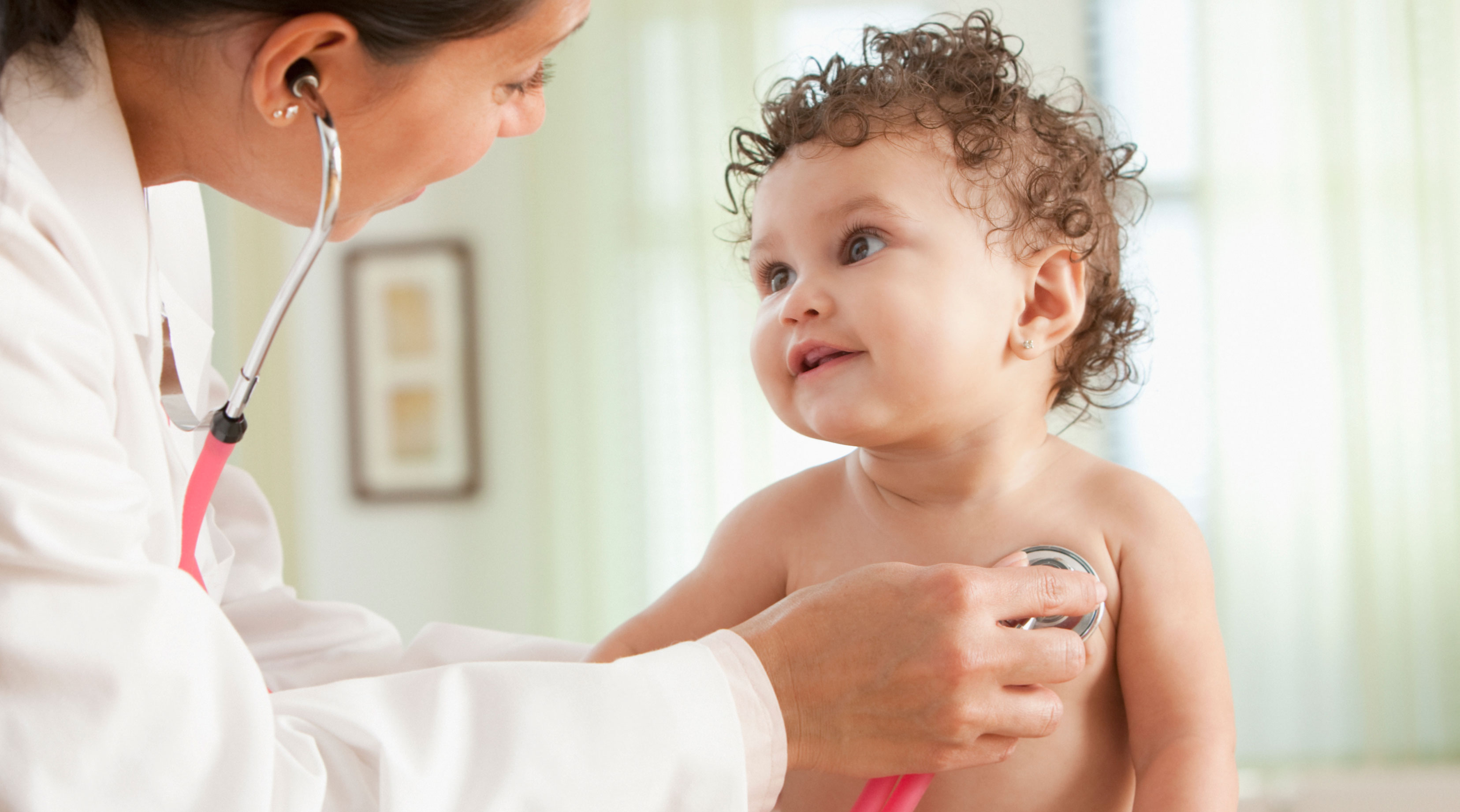
Moderna will begin testing its COVID-19 vaccine in kids younger than 12 years old—including babies as young as six months old.
The company announced the study, called the KidCOVE study, on Tuesday, March 16, citing plans to enroll 6,750 healthy kids in the U.S. and Canada. However, the company has not said how many kids are enrolled or have received their first dose so far.
According to their statement, the testing will occur in two parts, and the first will include kids aged 2 to 12. They will get two doses of 50 or 100 micrograms each 28 days apart. (For reference, an adult dose is 100 micrograms.) For kids younger than 2, the dosage will be 25, 50 or 100 micrograms. The first participants in the study will get the lowest doses and then will be monitored, Moderna said, while later participants will get higher doses. Researchers will then conduct an analysis to see which dosage is the safest and most effective for each age group.
In part two, kids will either get the dosage researchers deemed effective and safe from their analysis, or they will get placebo shots of salt water. All kids will be followed for a year after the study. Researchers will look at any side effects, their antibody levels and analyze any future COVID-19 infections, both with and without symptoms.
Moderna has also started trials of its vaccine in kids aged 12 to 17 and hope to have results by the summer. It’s important to note, however, that even with good results from the study the vaccine would first require authorization for use before it would be available.
In December, the American Academy of Pediatrics called for kids to be included in COVID-19 vaccine trials. While Moderna has already announced its plans to start trials in babies and young kids, Pfzier and BioNTech are currently testing its vaccine in kids aged 12 to 15. Johnson & Johnson also plans to test its vaccine in younger children after first testing it in older kids.
Please note: The Bump and the materials and information it contains are not intended to, and do not constitute, medical or other health advice or diagnosis and should not be used as such. You should always consult with a qualified physician or health professional about your specific circumstances.
Navigate forward to interact with the calendar and select a date. Press the question mark key to get the keyboard shortcuts for changing dates.