The FDA Has Approved the First-Ever Treatment for Peanut Allergies
Peanut allergies can be challenging because they’re often hard to predict. The smallest amount of exposure could cause severe reactions. But, now, the FDA has approved the first-ever treatment for peanut allergies in kids.
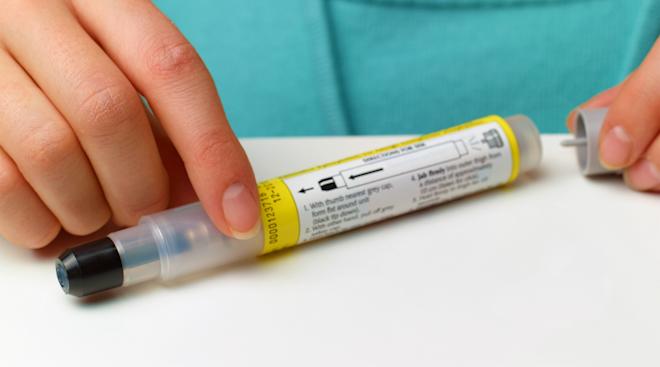
The news, which was announced on Friday, centers around a drug called Palforzia. It comes from Aimmune Therapeutics, who have been working on getting FDA clearance for almost two years. While the drug is not a cure, it is one step forward in reducing the amount of reactions kids have. Palforzia will target children aged 4 to 17 who already have a diagnosis for peanut allergies and actively avoid them. It’s important to note that Palforzia aims to help treat reactions from accidental exposure and should be used in emergency cases, the way you would an EpiPen.
“Peanut allergy affects approximately 1 million children in the U.S. and only 1 out of 5 of these children will outgrow their allergy. Because there is no cure, allergic individuals must strictly avoid exposure to prevent severe and potentially life-threatening reactions,” Peter Marks, M.D., Ph.D. and director of the FDA’s Center for Biologics Evaluation and Research stated in a news release. “Even with strict avoidance, inadvertent exposures can and do occur. When used in conjunction with peanut avoidance, Palforzia provides an FDA-approved treatment option to help reduce the risk of these allergic reactions in children with peanut allergy.” The drug, which is essentially a power made from peanuts, is designed to be administered in three phases over time. The first phase, called Initial Dose Escalation, lasts just one day, and the patient must consume the color-coded capsule under the supervision of a medical professional.
The second phase, called Up-Dosing, lasts for several months and moves through 11 increased dosage levels. Again, the first dose here should be taken under the supervision of a medical professional. According to the FDA, patients will be at highest risk for anaphylaxis, the most severe and possibly fatal allergic reaction, for these first two doses. If the dosage is received well by the patient, they can take the drug home to continue treatment.
The third phase is called Final Maintenance. In this phase, the patient takes a daily dose, now in the form of a sachet, by mixing the powder in semi-soft foods, such as applesauce or yogurt to consume. To combat the risk of anaphylaxis associated with the drug, the FDA is only giving treatment to patients (or who’s caregivers are) enrolled in the Risk Evaluation and Mitigation Strategy (REMS) program. It’s also requiring the healthcare providers giving the drug to be education on identifying and managing anaphylaxis.
Palforzia’s effectiveness was tested in multiple randomized, double-blind, placebo-controlled study of approximately 500 people. Researchers looked at what percent of the participants were able to tolerate a single 600 mg dose of peanut protein and only mild allergic symptoms after maintenance treatment. They found that 67.2 percent of participants were able to tolerate, as compared to only 4 percent of the placebo recipients. However, the studies did find that side effects to Palforzia may include abdominal pain, vomiting, nausea, tingling in the mouth, itching (including in the mouth and ears), cough, runny nose, throat irritation and tightness, hives, wheezing, shortness of breath and anaphylaxis. The FDA advises people with uncontrolled asthma against taking the drug.
While this may not be a cure, it is a step in the right direction for kids who continue to suffer from peanut allergies.
Please note: The Bump and the materials and information it contains are not intended to, and do not constitute, medical or other health advice or diagnosis and should not be used as such. You should always consult with a qualified physician or health professional about your specific circumstances.
Navigate forward to interact with the calendar and select a date. Press the question mark key to get the keyboard shortcuts for changing dates.