Groundbreaking Peanut Allergy Drug Could Save Lives
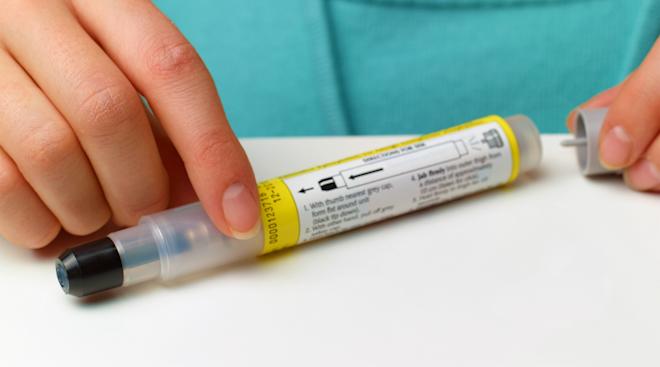
The results of a clinical experiment on peanut allergies was announced at a conference of the American College of Allergy, Asthma & Immunology in Seattle, and, if approved, it could be a gamechanger for kids with peanut allergies, The New York Times reports.
The year-long trial used an oral immunotherapy regimen aimed to reduce children’s sensitivity to peanut allergies by gradually exposing them to peanut protein over a six-month period. The experiment started with tiny amounts, which were carefully increased with doctors’ approval once a tolerance began to develop. The treatment is not meant to cure the allergy or allow children to eat foods with peanuts in them, but to reduce the risk of a life-threatening reaction due to accidental exposure to trace amounts of peanut allergens.
The study tested the treatment on 370 kids. After six months of treatment followed by six more months of maintenance therapy, two-thirds of the children were able to consume two peanuts without developing allergic symptoms. To put it in perspective, 124 children were given placebo powder and only 4 percent were able to consume the same amount of peanut without reacting.
But the treatment is not for everyone. Despite its great success, 20 percent of the kids who received treatment had to leave the study due to harmful side effects, compared to the less than 1 percent of kids who left that were on placebo. Plus, 14 percent of kids taking part in the treatment needed an emergency injection, compared to only 6.5 percent of those on placebo. Additionally, the treatment was not effective for adults who were tested.
Experts say the outcome exceeded their expectations, even calling the results “potentially lifesaving.” That said, they reinforce that the treatment does not cure peanut allergies and should not be attempted at home.
The study’s authors plan to submit a biologics license application to the FDA by the end of this year. The drug has already been designated as a breakthrough therapy, and, as a result, will receive priority review. It will go through an accelerated approval process, meaning the drug could be on the market by the end of 2019, The New York Times reports. While it hasn’t even been approved yet, the demand is expected to be high, and many are already expressing concern about how the drug will be priced and whether it will be covered by insurance.
For parents of kids with peanut allergies, this could help bring piece of mind. Until then, learn about the Nima Peanut Sensor, a tiny machine which tests samples of food for traces of peanuts.
Please note: The Bump and the materials and information it contains are not intended to, and do not constitute, medical or other health advice or diagnosis and should not be used as such. You should always consult with a qualified physician or health professional about your specific circumstances.
Navigate forward to interact with the calendar and select a date. Press the question mark key to get the keyboard shortcuts for changing dates.