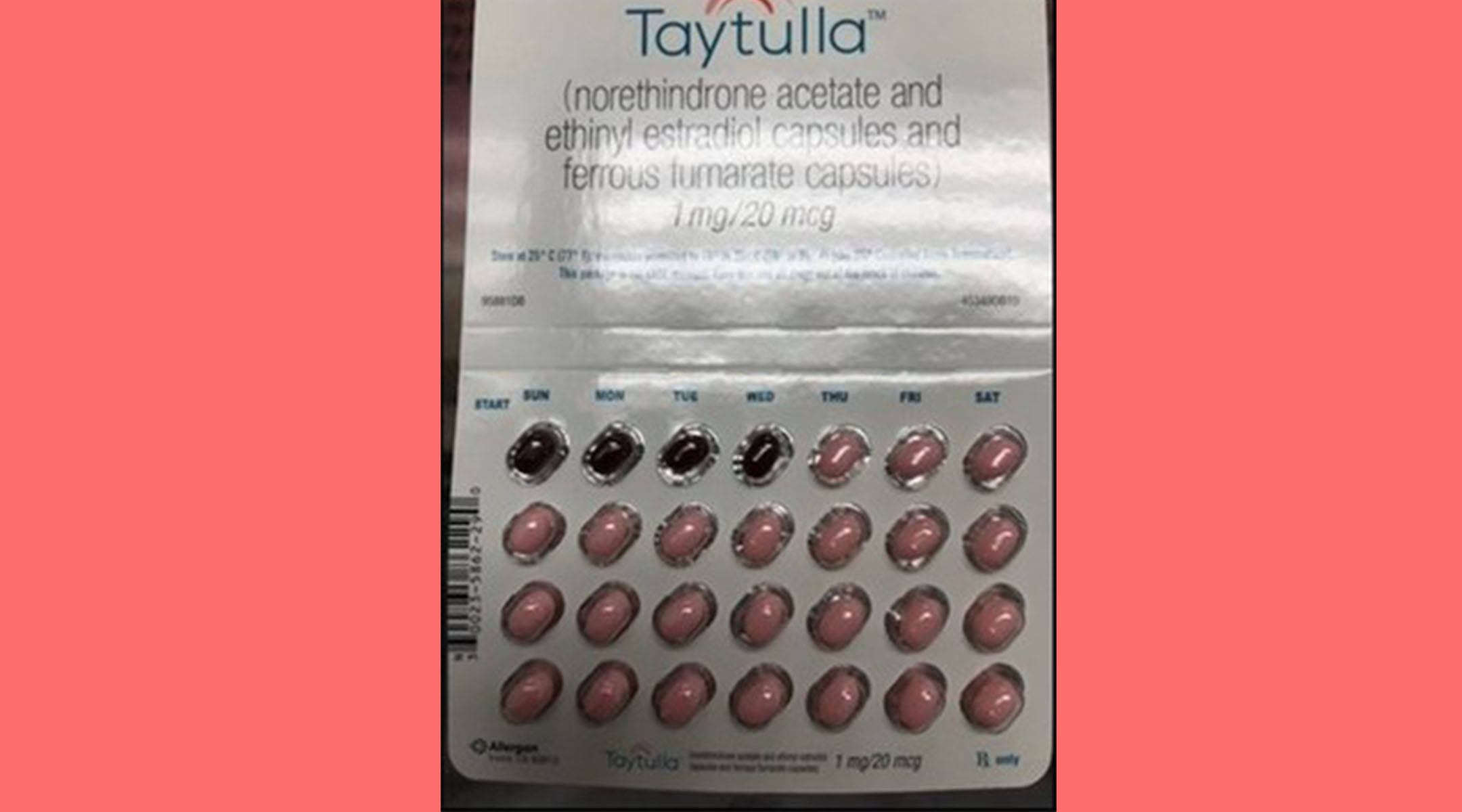
Birth control after baby? While OBs prefer you go with an IUD, the pill continues to be the most popular birth control method in the US. If you’re one of the 9.7 million women on the pill, consider taking a close look at this month’s pack. Sample packs of Taytulla, manufactured by Allergan, are being recalled for a packaging error.
In the affected packages, the placebo capsules are incorrectly placed in the first four days instead of active capsules. Taytulla should start with 24 active pink capsules and end with four inactive maroon capsules. As you probably know, taking oral contraceptives out of order can lead to unintended pregnancy.
“The reversing of the order may not be apparent to either new users or previous users of the product, increasing the likelihood of taking the capsules out of order,” reads a statement from Allergen. “If you are a patient in the US who has used a Taytulla sample pack from lot 5620706, Exp. May 2019 since August 27, 2017, and are concerned that you may be impacted by this issue, please consult with your physician.”
Allergen adds that thanks to inspections on the production line, only one lot of Taytulla is affected.
Navigate forward to interact with the calendar and select a date. Press the question mark key to get the keyboard shortcuts for changing dates.